Romania marks a significant milestone in the management of migraine with the reimbursement of the full class of anti-CGRP (Calcitonin Gene-Related Peptide) therapies for migraine prevention in adult patients presenting with more than four migraine days per month, effective March 2026.
Anti-CGRP Therapies: A New Standard of Care Now Accessible
The following treatments have been approved and reimbursed under the national health system:
Monoclonal antibodies targeting the CGRP pathway:
- Galcanezumab
- Fremanezumab
- Eptinezumab
Small molecule CGRP receptor antagonist (gepant class):
- Rimegepant — also indicated for migraine prevention
This decision was approved by the Ministry of Health and the National Health Insurance House and published in the Monitorul Oficial al României, the country’s official gazette, conferring full legal standing to the reimbursement protocol.
It is worth noting that Galcanezumab had already been approved in Romania for migraine prevention in adults since December 2022, making it a pioneer in this class. The 2026 expansion brings the entire anti-CGRP class under reimbursement, ensuring broader and more equitable patient access.
Preventive Treatment Landscape: Traditional Options Remain Available
Alongside the newly reimbursed therapies, traditional preventive treatments remain accessible at affordable costs, including:
- Topiramate and Valproic acid — available, though currently reimbursed only under the epilepsy indication
- Beta-blockers
- Candesartan
It should be noted that Botulinum toxin is not currently registered for migraine in Romania; its approved indications remain limited to dystonia and spasticity.
Acute Migraine Treatment: Affordable Triptans Now Available
Romania also offers improved access to acute migraine therapies at affordable prices:
- Sumatriptan (oral and subcutaneous 3 mg) — subcutaneous formulation is reimbursed via general practitioners
- Rizatriptan and Frovatriptan — introduced in 2025 at accessible price points
It is hoped that Romania will be included in the European Map of Access to Migraine Treatment in recognition of these meaningful advances in migraine care.
Bibliography & References
- Monitorul Oficial al României, Partea I, Nr. 175 bis/6.III.2026 — Protocol terapeutic corespunzător poziției nr. 29 cod (N02CD): Migrena: Galcanezumabum, Fremanezumabum, Eptinezumabum, Rimegepant. Ministry of Health and National Health Insurance House of Romania, 2026.
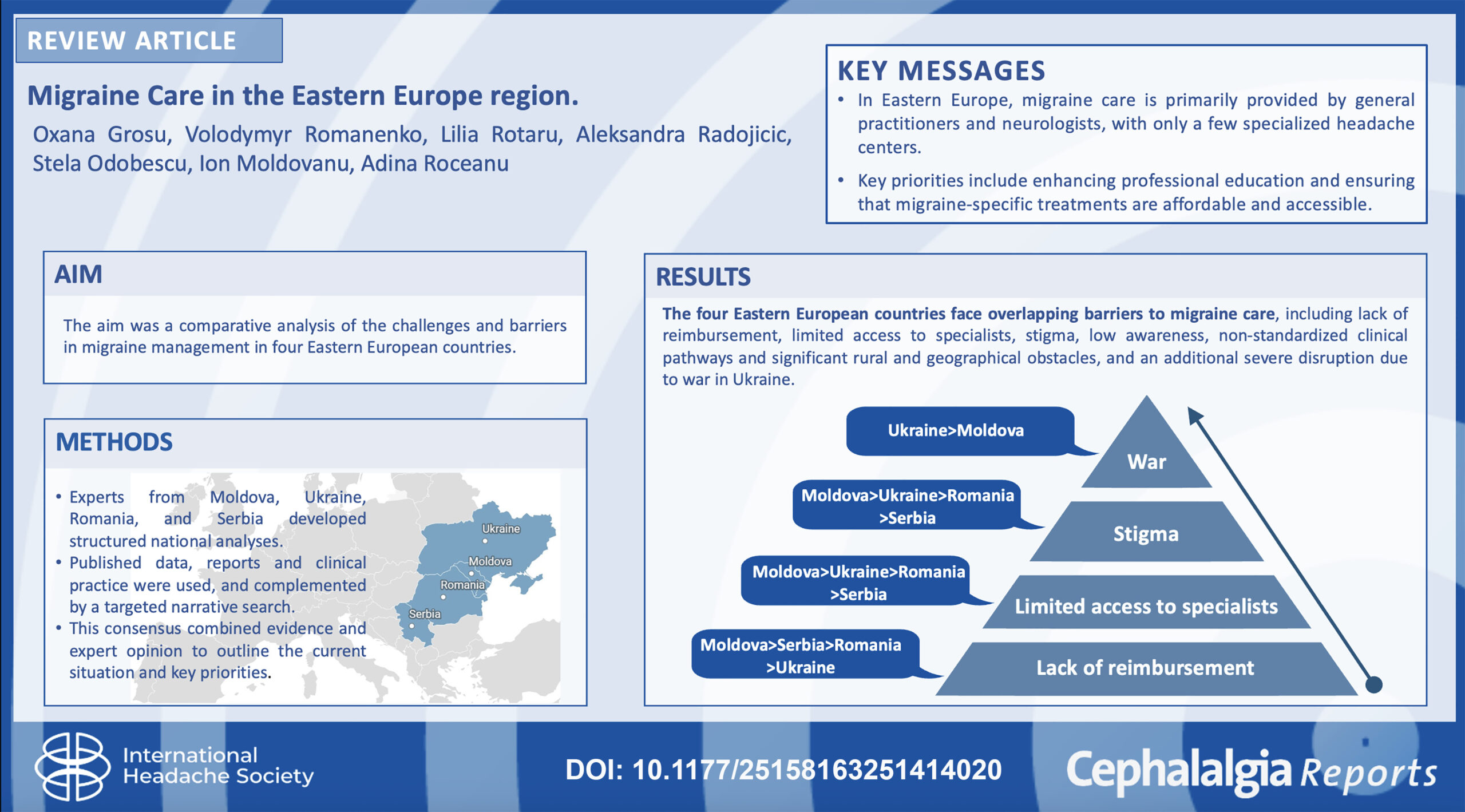
- Grosu O, Romanenko V, Rotaru L, Radojicic A, Odobescu S, Moldovanu IV, Roceanu A. Migraine care in the Eastern Europe region. Cephalalgia Reports. 2026;9:1–13. DOI: 10.1177/25158163251414020
- European Migraine & Headache Alliance (EMHA) — European Map of Access to Migraine Treatment. Available at: https://www.emhalliance.org/european-map/








